Rationale
Quetiapine is an antipsychotic medication approved for the treatment of schizophrenia, acute mania episodes, bipolar disorder, and as an adjunctive treatment for major depressive disorder. Patients on quetiapine are at risk of experiencing QTc prolongation. 1 The QT interval is a representation of the duration of the ventricular action potential on electrocardiogram; however, this interval needs to be corrected for heart rate. This correction is known as QTc. The QTc interval is prolonged when it is greater than 440 ms in men or greater than 460 ms in women. This prolongation can be life threatening if the interval extends past 500 ms, putting patients at risk for developing torsades de pointes.2 If left untreated, it can be fatal.3As monotherapy, patients may not experience this adverse effect, but additional factors such as a concomitant QT prolonging agent or inhibition of the metabolism of quetiapine can increase the risk of this effect during treatment.4
Kratom (Mitragyna speciosa) is a tropical tree indigenous to Southeast Asia and has garnered attention for its suggested utility in pain management, alleviation of opioid withdrawal, and anxiety modulation, owing to its dual stimulant and opioid-like properties. The manifestation of these effects is contingent upon dosage, with stimulant attributes prevailing at lower doses and opioid effects dominating at higher doses. The active constituents responsible for these effects are mitragynine and 7-hydroxymitragynine.5,6 Although utilization has been predominantly in Asia, the popularity of kratom has recently surged in the United States. The American Kratom Association estimates that 10-16 million individuals employ kratom as an alternative to opioids, for recreational purposes, and to mitigate opioid withdrawal symptoms.7 The escalating popularity of this natural product has prompted its inclusion in the US Drug Enforcement Administration’s (DEA) “Drugs of Concern” list, attributing this designation to its sedative properties and perceived addiction potential; however, this was later retracted due to public outcry.8
While kratom raises potential safety apprehensions linked to pharmacodynamic interactions with specific prescription medications, the primary focus of this investigation pertains to its involvement in pharmacokinetic interactions with prescription drugs.9 Ref Research highlights the ability of kratom to hinder the metabolism of specific medications, potentially resulting in heightened blood concentrations and consequential severe adverse effects.10 Consequently, a thorough examination of the evidence concerning kratom’s potential metabolic inhibitory effects becomes imperative to mitigate the risk of interactions between kratom and quetiapine.
Algorithm
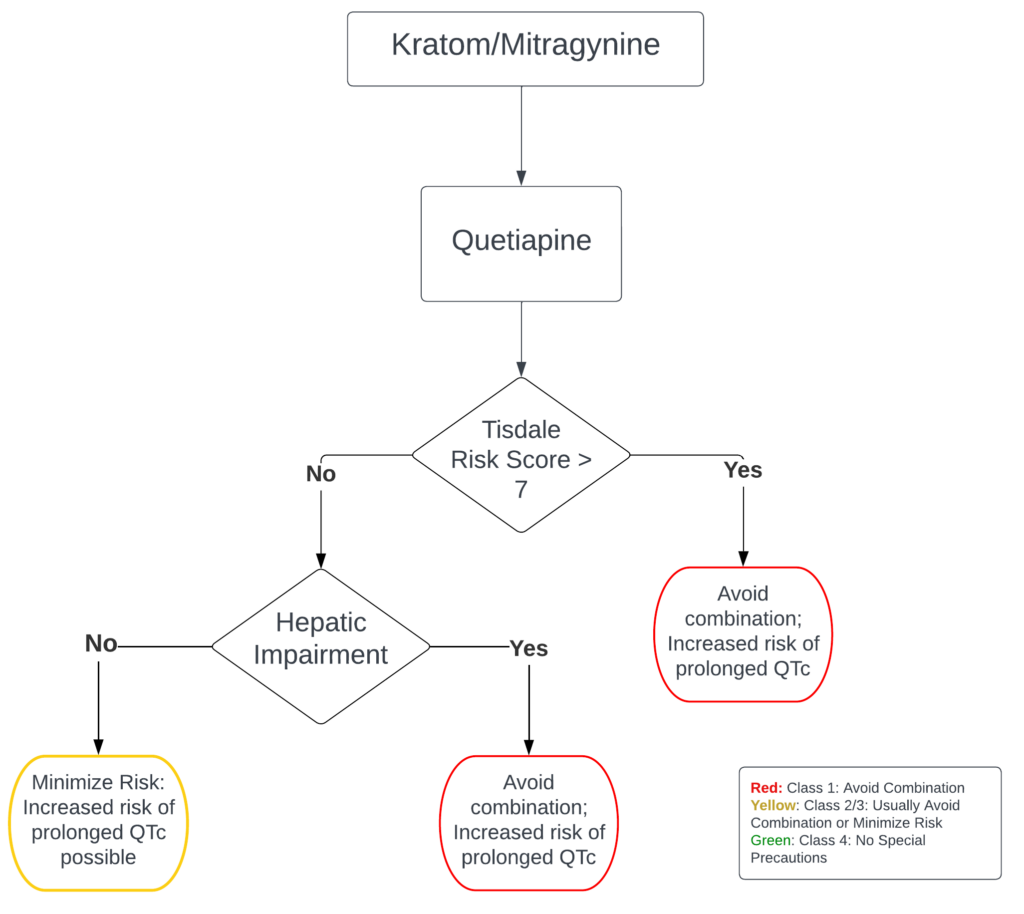
Explanation
Quetiapine is primarily eliminated through hepatic metabolism and more specifically by cytochrome P450 (CYP) 3A4.4 As a result, concomitant use of CYP3A4 inhibitors can lead to an increase in quetiapine concentrations that may require a dose reduction. In the context of its interaction with kratom, the natural product has the potential to inhibit the metabolism of CYP3A4. This interaction can lead to a significant adverse effect such as QTc prolongation due to increased quetiapine concentration.4,11,12 Hence, it is important to understand the supporting evidence for kratom NPDIs presented from metabolic studies and case reports in the literature.
A study published in 2020 investigated the effects of mitragynine on three major cytochrome P450s, specifically CYP2C9, CYP2D6, and CYP3A4. Mitragynine exhibited concentration-dependent inhibition of all three enzymes, with the most pronounced effect observed for CYP2D6.13 A 2021 study showed similar results, as mitragynine and three kratom extracts displayed concentration-dependent inhibition of CYP2C9, CYP2D6, and CYP3A in human liver microsomes (HLMs). At the minimum tested concentration (2 μg/mL), the kratom extracts inhibited CYP2D6, CYP2C9, and CYP3A by 44% to 64%, 24% to 29%, and 15% to 23%, respectively. Mitragynine at its lowest tested concentration (1 μM) inhibited the same three enzymes (CYP2D6, CYP2C9, and CYP3A) by 57%, 21%, and 26%, respectively. In addition to testing concentration inhibition in HLMs, the three kratom extracts along with mitragynine were also evaluated for their inhibition potential in human intestinal microsomes (HIMs). The study found the kratom extracts at their lowest tested concentrations inhibited CYP3A by 24-25% and 9%, respectively. Lastly, the study found two interesting results from IC50 shifts experiments they conducted. The first is that mitragynine was found to be a strong competitive inhibitor of CYP2D6; the second was that mitragynine showed time dependent inhibition of CYP3A activity. Hanapi et al. reported similar trends showing that mitragynine inhibited CYP2D6, CYP2C9, and CYP3A4, with IC50 values of 0.45±0.33 mM, 9.70±4.80 μM and 41.32±6.74 μM, respectively.14
There are two case reports that have been identified displaying the potential for kratom to inhibit CYP metabolism.15,16 The first case report details a 36-year-old Caucasian man who presented to the emergency department for dizziness. At the time, he only reported taking venlafaxine 150 mg daily and quetiapine 300 mg nightly; however, it was later discovered he had taken kratom as a way to treat his substance use disorder. At the time of the visit, evaluation was notable for high blood pressure (144/92 mm Hg), tachycardia (116 bpm), and elevated temperature (99.5 °F) and an electrocardiogram showing sinus tachycardia and a prolonged QTc (563 msec). The patient was advised to discontinue venlafaxine and quetiapine due to concern for serotonin syndrome and prolonged QTc.10 Proposed mechanisms as stated by the case report include inhibition of CYP2D6 and CYP3A causing supratherapeutic concentrations of venlafaxine and quetiapine since these drugs are substrates of CYP2D6 and CYP3A respectively in addition to potential pharmacodynamic interactions caused by kratom’s physiological effects.4,15 Additionally, a normal QTc interval is less than 450 ms for men and less than 460 ms for women indicating a prolonged QTc interval for this patient.2
Another case report involved a 27-year-old male, previously diagnosed with Asperger Syndrome, bipolar disorder, and a history of substance abuse, was found deceased in his residence. The decedent had multiple prescription medications in his residence at the time of death, and a postmortem forensic toxicology report indicated valproic acid was quantitatively positive at 8.8 mcg/mL, quetiapine was quantitatively positive at 12000 ng/mL, and mitragynine was qualitatively positive. The cause of death for the decedent was ruled as acute toxic effects of quetiapine complicated by mitragynine use, and the death was classified as an accident. This conclusion is consistent with the reference ranges for both quetiapine and valproic acid, which are 100 to 500 ng/mL and approximately 50 to 100 mcg/mL, respectively. Similar to the previous case report, a proposed mechanism was that quetiapine metabolism by CYP3A was inhibited by mitragynine.11,16,17 The potential risk associated with QT prolongation involving quetiapine and kratom becomes more nuanced when considering the interaction with risk factors for QT prolongation. Tisdale et al. developed a risk score for determining the risk a patient has for developing QT prolongation. Some of the risk factors considered include sex and age of patient, presence of comorbidities such as MI or left ventricular dysfunction, and use of 2 or more QT prolonging agents.18 The risk of using two or more QT prolonging agents becomes a interesting discussion point as recent studies have suggested that using more than one QT prolonging agent does cause a cumulative effect as one would expect. A recent 2022 study delving into the impact of hydroxychloroquine on the QTc interval, both in isolation and in combination with other QT-prolonging agents showed that the addition of a QT prolonging agent did not exert a significant influence on the patient’s QTc interval. Multiple studies have found similar results; however, further comprehensive studies are necessary to either substantiate or challenge this finding.19 Based on the above case studies and in vitro studies, kratom/mitragynine should be used with caution for patients taking quetiapine.
Artifacts for implementers
We have created a prototype CDS application as a SMART app – see our experimental apps page
References
- Maan JS, Ershadi M, Khan I, et al. Quetiapine. [Updated 2023 Aug 28]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459145/
- Rezuş C, Moga VD, Ouatu A, Floria M. QT interval variations and mortality risk: is there any relationship? Anatol J Cardiol. 2015 Mar;15(3):255-8. doi: 10.5152/akd.2015.5875. PMID: 25880179; PMCID: PMC5337065.
- Crawford MH, Aras M, Sanchez JM. eds. Torsades de Pointes (https://accesscardiology.mhmedical.com/content.aspx?bookid=2470§ionid=196389720). Quick Dx & Rx: Cardiology. McGraw Hill. Accessed 2/10/2024.
- DeVane CL, Nemeroff CB. Clinical pharmacokinetics of quetiapine: an atypical antipsychotic. Clin Pharmacokinet. 2001;40(7):509–522. [PubMed: 11510628]
- Garcia-Romeu A, Cox DJ, Smith KE, Dunn KE, Griffiths RR. Kratom (Mitragyna speciosa): User demographics, use patterns, and implications for the opioid epidemic. Drug Alcohol Depend. 2020 Mar 1;208:107849. doi: 10.1016/j.drugalcdep.2020.107849. Epub 2020 Feb 3. PMID: 32029298; PMCID: PMC7423016.
- Prozialeck WC, Jivan JK, Andurkar SV. Pharmacology of kratom: an emerging botanical agent with stimulant, analgesic and opioid-like effects. J Am Osteopath Assoc. 2012;112(12):792–799. [PubMed: 23212430]
- Henningfield JE, Grundmann O, Babin JK, Fant RV, Wang DW, Cone EJ. Risk of death associated with kratom use compared to opioids. Prev Med. 2019 Nov;128:105851. doi: 10.1016/j.ypmed.2019.105851. Epub 2019 Oct 21. PMID: 31647958.
- Schimmel J, Amioka E, Rockhill K, Haynes CM, Black JC, Dart RC, Iwanicki JL. Prevalence and description of kratom (Mitragyna speciosa) use in the United States: a cross-sectional study. Addiction. 2021 Jan;116(1):176-181. doi: 10.1111/add.15082. Epub 2020 Apr 28. PMID: 32285981.
- Tanna RS, Tian DD, Cech NB, et al. Refined prediction of pharmacokinetic kratom-drug interactions: time-dependent inhibition considerations. J Pharmacol Exp Ther. 2021;376(1):64–73. [PubMed: 33093187]
- Tanna RS, Cech NB, Oberlies NH, Rettie AE, Thummel KE, Paine MF. Translating Kratom-Drug Interactions: From Bedside to Bench and Back. Drug Metab Dispos. 2023 Aug;51(8):923-935. doi: 10.1124/dmd.122.001005. Epub 2023 Jun 7. PMID: 37286363; PMCID: PMC10353077.
- Lexicomp. (n.d.). Quetiapine: Drug information. UpToDate. Retrieved January 8, 2024, from https://www-uptodate-com.pitt.idm.oclc.org/contents/quetiapine-drug-information?search=quetiapine&source=panel_search_result&selectedTitle=1~136&usage_type=panel&kp_tab=drug_general&display_rank=1#F55476430
- Warner ML, Kaufman NC, Grundmann O. The pharmacology and toxicology of kratom: from traditional herb to drug of abuse. Int J Legal Med. 2016;130:127–38.
- Todd DA, Kellogg JJ, Wallace ED, et al. Chemical composition and biological effects of kratom (Mitragyna speciosa): in vitro studies with implications for efficacy and drug interactions. Sci Rep. 2020;10(1):19158. [PubMed: 33154449]
- Hanapi NA, Ismail S, Mansor SM. Inhibitory effect of mitragynine on human cytochrome P450 enzyme activities. Pharm Res. 2013;5:241–6.
- Brogdon HD, McPhee MM, Paine MF, Cox EJ, Burns AG. A Case of Potential Pharmacokinetic Kratom-drug Interactions Resulting in Toxicity and Subsequent Treatment of Kratom Use Disorder With Buprenorphine/Naloxone. J Addict Med. 2022;16(5):606-609. doi:10.1097/ADM.0000000000000968
- Hughes RL. Fatal combination of mitragynine and quetiapine – a case report with discussion of a potential herb-drug interaction. Forensic Sci Med Pathol. 2019;15(1):110-113. doi:10.1007/s12024-018-0049-9
- Lexicomp. (n.d.). Valproate: Drug information. UpToDate. Retrieved January 8, 2024, from https://www-uptodate-com.pitt.idm.oclc.org/contents/valproate-drug-information?search=valproic%20acid&source=panel_search_result&selectedTitle=1~148&usage_type=panel&kp_tab=drug_general&display_rank=1#F232992
- Tisdale JE, Jaynes HA, Kingery JR, Mourad NA, Trujillo TN, Overholser BR, Kovacs RJ. Development and validation of a risk score to predict QT interval prolongation in hospitalized patients. Circ Cardiovasc Qual Outcomes. 2013 Jul;6(4):479-87. doi: 10.1161/CIRCOUTCOMES.113.000152. Epub 2013 May 28. Erratum in: Circ Cardiovasc Qual Outcomes. 2013 Nov;6(6):e57. PMID: 23716032; PMCID: PMC3788679.
- Villa Zapata L, Boyce RD, Chou E, et al. QTc Prolongation with the Use of Hydroxychloroquine and Concomitant Arrhythmogenic Medications: A Retrospective Study Using Electronic Health Records Data. Drugs Real World Outcomes. 2022;9(3):415-423. doi:10.1007/s40801-022-00307-5
Authorship
Author: Dr. Kojo Abanyie with input from Dr. Daniel Malone, Dr. Mary Paine, and Dr. Ainhoa Gomez Lumbreras
Email: koa27@pitt.edu
Date: March 25, 2024